- What Are Some Advantages And Disadvantages Of The Serial Dilution Agar Plate Technique
- 6. What Are Some Advantages And Disadvantages Of The Serial Dilution Agar Plate Technique
Each calibration standard solution is prepared based on the previous calibration standard. The process involves taking a portion of the previous standard and diluting it with the solvent to obtain the next calibration standard. The errors introduced with each successive dilution drops proportionately with the solution concentration. Preparing a series of calibration standards by this method reduces the amount of required time. Most calibration standards span a large range of concentrations, so the accuracy of the calibration standard prepared increases. Calibrations Solutions More Evenly Spaced.
The dilution factor chosen for the series of calibration standards is achievable by using serial dilution. The progression of calibration standard concentration is always a geometric series. Consider the example of making the first standard at 1/3 the concentration of the known, the next calibrant would be 1/9th the concentration of the known and the following two calibrants formed are 1/27th and 1/81st. This becomes a much greater advantage when the span of the calibration standards must cover several orders of magnitude in concentration.
The Quadrajet (Q-Jet) carburetor is better on a street/daily driven vehicle for a couple reasons. One is that the primary throttle bores are pretty small, so at part throttle, like most driving, you won't be using the entire carb, and hence you'll use less gas. Take for example a mechanical secondary Holley four barrel. At part throttle, all four barrels are open and spraying fuel. In a Q-Jet, only two barrels are open and spraying fuel. One other reason is that it's a vacuum secondary carb. This means that the secondaries open a bit slower, so if you need to gun it, you'll have the power, but won't use all the fuel at once, saving you a bit.
Again, take a Holley for example. Go from 25% throttle to 100%. You go from four barrels spraying some fuel to all barrels spraying maximum capacity.
This could bog your engine down from too much fuel for too few RPMs if not tuned correctly, and will immediately use more fuel. On the other hand, the Q-Jet would open the secondaries slower, allowing the engine to come up into the higher RPMs it would need for the extra fuel. Say you go from 25% to 100% throttle. You go from two barrels spraying some fuel to two barrels spraying maximum capacity, and then in addition, two bigger barrels spraying 25% capacity, 50% capacity, 75% capacity and if you stay in it long enough, 100% capacity. This would use progressively more and more fuel, so for a quick spurt, you would save some fuel you would have used with the Holley, say only going up to 50% capacity on the secondaries compared to the Holleys 100% capacity immediately. But, you do lose some responsiveness.
This all still happens pretty quickly, as in within a split second for the Holley to go 100% compared to a Q-Jet taking 1 second or so to go fully open. Other than that, the Q-Jet is basically on par with other carbs. They can be harder to tune, but if you know what you're doing, it will just take a bit more time and effort, not much.
In a cruiser or daily driver, you'd be better off using a Q-Jet. For a race car or weekend toy, just go for the Holley since you won't be driving it all that much, and they are easier to set up and tune for most people.
Pacific, North American, South American, Eurasian, Persian subplate, African, Somalian Subplate, Austral-Indian, Antarctic Technically they are referred to as tectonic plates, between the 7 major tectonic plates they cover most of the earth's surface except for where a few smaller ones exist. Because they cover such a large area the borders of each plate won't correspond to any specific counrty or reference point that can easily be referred to as a particular plates location. Extremely generalised locations are stated below for the 7 largest techtonic plates. Pacific Plate - covers the majority of the Pacific Ocean 2.
Antarctic Plate - covers Antarctica and much of the far Southern Ocean area 3. Indo-Australian Plate - stretches through Indonesia, Australia, New Zealand, Papua New Guinea and on to the Canary Islands 4. Eurasian Plate - covers all of Europe and most of Asia except the Philippines 5. African Plate - covers Africa and much of the surrounding ocean 6. North America plate - Covers North America and Arctic regions 7.
South America Plate - covers South America and a large portion of the southern Atlantic Ocean There are eight smaller plates that are still larger than most countries.
Part C: UV Experiments Serial Dilutions and Viable Cell Counts The experiment Observing the Effects of Solar Ultraviolet Radiation on Cells shows that when cells are exposed to sunlight all, some, or none of them may be killed. Many experimental questions can be answered with qualitative answers like 'all, some, or none.' Other questions may require quantitative answers. For example, in the next experiment you will use the sensitive yeast strain to measure the intensity of solar UV radiation by measuring the fraction of cells exposed that survive. To get quantitative answers about yeast survival you must put a known numbers of viable (living) cells onto the agar plates and then count the number that remain after being exposed. You can determine the number of viable cells by counting the colonies that grow up on the agar growth medium in a Petri plate by assuming that each colony grows from a single viable cell.
This is usually a reasonable assumption. Experiment: In the experiment that follows you will learn how to measure the number of viable cells on a Petri plate.
Hexcmp2 2 34 serial number. You will be able to use this procedure whenever you need to measure the number of cells that survive an exposure to radiation or some other treatment. First you will estimate the number of cells in a liquid suspension in order to plate a reasonable number of cells. For this you will use one of the most sophisticated and sensitive optical instruments in existence, the human eye.
With surprisingly little practice you can learn to estimate the number of cells in a suspension by just looking at it. You can estimate cell density because of your eyes' fairly sharp threshold for observing turbidity (cloudiness). When viewed in a standard 13 100 mm glass tube, yeast suspensions of less than about 1 million cells per mL are not visibly turbid. Above this threshold density, the suspension is cloudy. When you adjust the number of cells in a suspension until just barely visible, you obtain a suspension of known density (approximately 1 106 cells/ml). When you have a suspension that contains approximately 1 106 cells/ml, you will dilute it to get the right concentration for plating.
You will make the dilutions in known steps so you can calculate the number of cells in each dilution tube. This procedure helps you plate a countable number of colonies.
With or without a filter, it's the exploit itself that counts, how can you not see that? /use 0 1 /run PickupContainerItem(0,1) if CursorHasItem() then PickupContainerItem(0,2) end /Click StackSplitOkayButton /Click ContainerFrame1Item16 Your logic is ****ing priceless. And for your information, maxi also gave a macro to some people who didn't want the exe. Wow item dupe hack 3.3.5. It's like saying 'Maxi's exploit worked with FF17, but here I'm using the FF18 slot so yeah it's not the same exploit so I can leak it.' And for your information, maxi also gave a macro to some people who didn't want the exe.
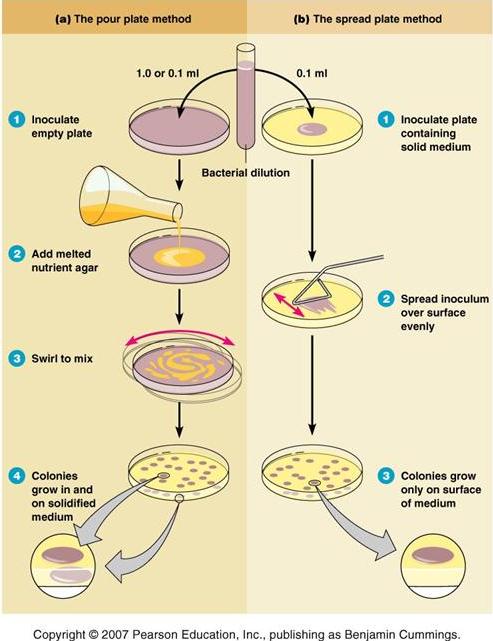
The number of microorganisms present in the particular test sample is determined using the formula: CFU/mL= CFU. dilution factor. 1/aliquot For accurate counts, the optimum count should be within the range of 30-300 colonies/plate. To insure a countable plate a series of dilutions should be plated.The pour plate method of counting bacteria is more precise than the, but, on the average, it will give a lower count as heat sensitive microorganisms may die when they come contact with hot, molten agar medium. Uses: The pour plate technique can be used to determine the number of microbes/mL in a specimen.
It has the advantage of not requiring previously prepared plates, and is often used to assay bacterial contamination of food stuffs. Materials and Equipments. Test sample. Plate Count Agar (PCA) or. Hot water bath 45°C. Sterile Petri dishes.
Flame. Colony counter with magnifying glass.
Sterile capped 16.150 mm test tubes. Pipettes of various sizes (e.g. 01, 1.0 and 2.0 mL) Procedure of Pour plate technique. Prepare the dilution of the test sample expected to contain between 30-300 CFU/mL. (Follow serial dilution technique). Inoculate labeled empty petri dish with specified mL (0.1 or 1.0 mL) of diluted specimen Note: for the detail description regarding use of pipette, inoculation of sample, dilution technique etc, follow the reference 1.
Pouring the molten agar and incubation. Collect one bottle of sterile molten agar (containing 15 mL of melted Plate Count Agar or any other standard culture media) from the water bath (45°C).

Pouring the molten agar medium. Hold the bottle in the right hand; remove the cap with the little finger of the left hand. Flame the neck of the bottle. Lift the lid of the Petri dish slightly with the left hand and pour the sterile molten agar into the Petri dish and replace the the lid. Flame the neck of the bottle and replace the cap. Gently rotate the dish to mix the culture and the medium thoroughly and to ensure that the medium covers the plate evenly. Do not slip the agar over the edge of the petri dish.
Allow the agar to completely gel without disturbing it, it will take approximately 10 minutes. Seal and incubate the plate in an inverted position at 37°C for 24-48 hours. Overview of Pour plate method and spread plate method Results: After 24-48 hours, count all the colonies ( again: note that the embedded colonies will be much smaller than those which happen to form on the surface). A magnifying colony counter can aid in counting small embedded colonies. Calculate CFU/mL using the formula: CFU/mL= CFU.
What Are Some Advantages And Disadvantages Of The Serial Dilution Agar Plate Technique
dilution factor. 1/aliquot (the volume of diluted specimen (aliquot) is either 0.1 or 1.0 mL) Disadvantages of Pour plate method. Preparation for pour plate method is time consuming compared with streak plate/and or spread plate technique.
Loss of viability of heat-sensitive organisms coming into contact with hot agar. Embedded colonies are much smaller than those which happen to be on the surface.
Thus, one must be careful to score these so that none are overlooked. Reduced growth rate of obligate aerobes in the depth of the agar. References and further readings. Basic Practical Microbiology A Manual by Society for General Microbiology (SGM).
Measurement of Microbial Growth: A number of techniques are available in order to measure growth of microbial populations. Ar rahman 5.1 songs free download. Either population number of mass may be calculated ad growth leads to increase in both. Direct measurement of cell numbers: Bacteria or microorganisms can be counted directly on the plate and also called as plate counting. Advantage of this method is that it measures the number of viable cells. Disadvantage is that, it is time consuming and expensive as one needs media and other conditions need to be maintained. Bacteria counted on plate counts are referred to as colony forming units as a single cell or a clump of bacterial cells can lead to a colony which contains many cells. The colonies when they are counted in plate count method are to be present sparsely for accurate counting as overcrowding can lead to incorrect counting.
To solve this, one has to adapt the serial dilution method in order to get an accurate count. Serial dilution and pour and spread plate: Supposing one has to accurately count the number of cells given in a solution, then serial dilution needs to be performed. A 1ml of the sample is taken and transferred to a tube containing 9ml of sterile water and this process can be repeated until we reach a considerable dilution (say 10 6 to 10 7). Once the original inoculum is diluted one needs to perform a pour plate or a spread plate technique in order to count the number of bacteria present in the diluted sample and then the original sample.
6. What Are Some Advantages And Disadvantages Of The Serial Dilution Agar Plate Technique
In pour plate method the diluted sample is poured into the petriplate and then the medium which is at nearly 50 0C is poured over the inoculum and mixed by gentle agitation. With this method, colonies grow within the nutrient agar as well as on the surface of the agar plate. As certain disadvantages are encountered in this method like heat sensitive microorganisms might not grow and also bacteria when they grow within the nutrient medium might not be useful for diagnostic purposes.
In order to avoid these problems, spread plate method is mostly used (Fig. A 0.1ml of the diluted sample is added to the surface of the nutrient medium and spread uniformly with the help of a glass spreader and after incubation, the colonies can be counted and the concentration of the bacterial cells in the original sample is calculated as follows: Number of bacteria/ml = Number of colonies on plate x reciprocal of dilution of sample.
